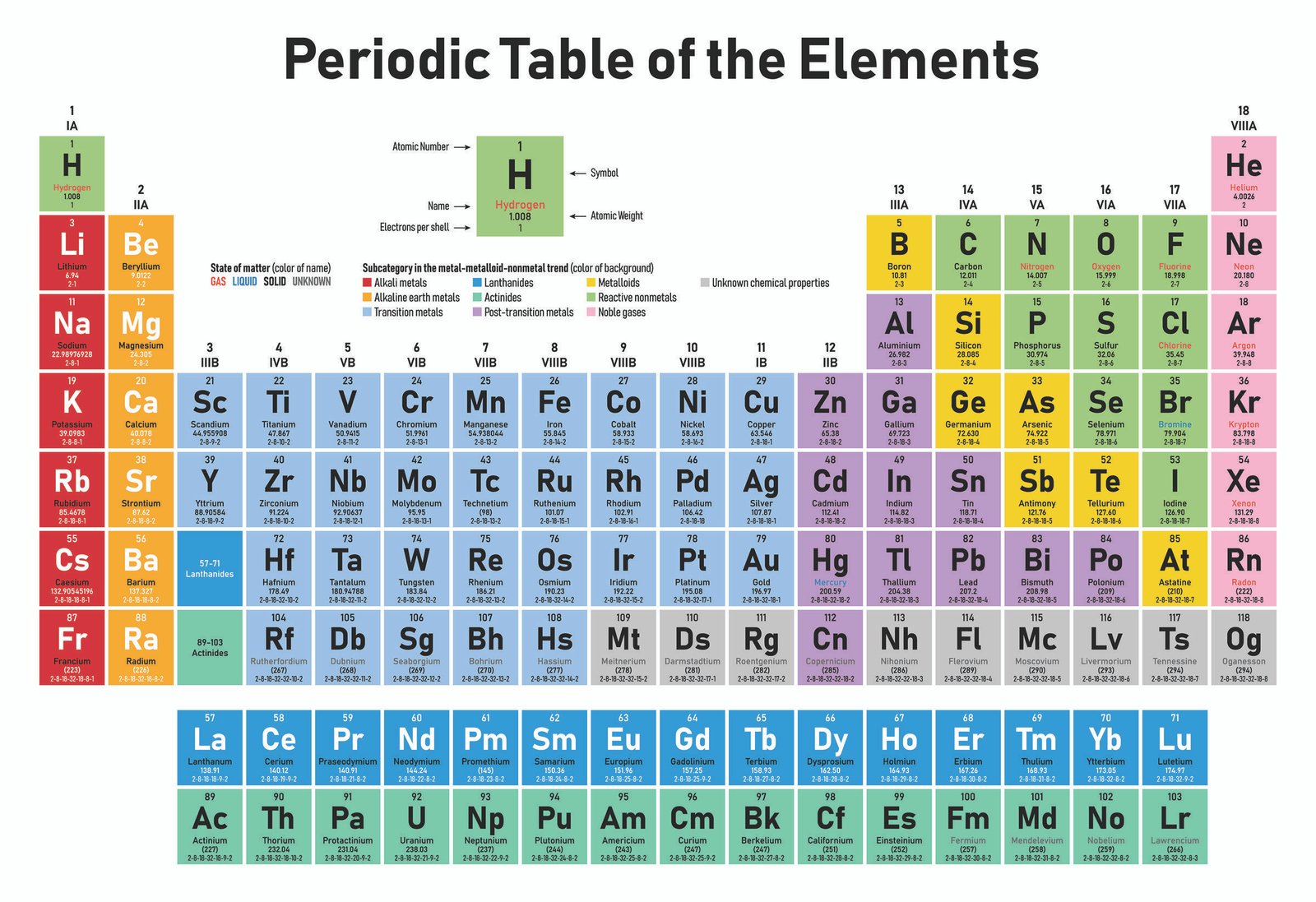
The atoms in a periodic table family or group (a vertical column ofĮlements) will all have the same valence (outer) electron configurations with respect to The arrangement of the atoms in the standard periodic table is based on the electronĬonfigurations. Periodic Trends in Electron Configurations Not concern ourselves with these in this review. The above general filling order and the observed electron configurations–particularlyįor the transition (d-block) elements and the inner transition (f-block) elements. Note that there are some discrepancies between the predicted configurations based on Number of electrons present in the atom or ion. Note that the sum of the numbers of electrons in each subshell is equal to the total Zr 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 2Įxample Problem: Write the electron configuration for the ground state of an atom ofĪs 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 For example, using the above filling order we would write theĮlectron configuration for the 10 electrons in neon as Ground state) of an atom or ion is the one in which all electrons are assigned to the 
Orbital can be assigned a maximum of two electrons. Subshell contains five d orbtials, each f subshell contains seven f orbitals, etc., and each Of the various electron-electron interactions and generally follow the orderġs < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f ≈ 5d <Įach s subshell contains one s orbital, each p subshell contains three p orbitals, each d 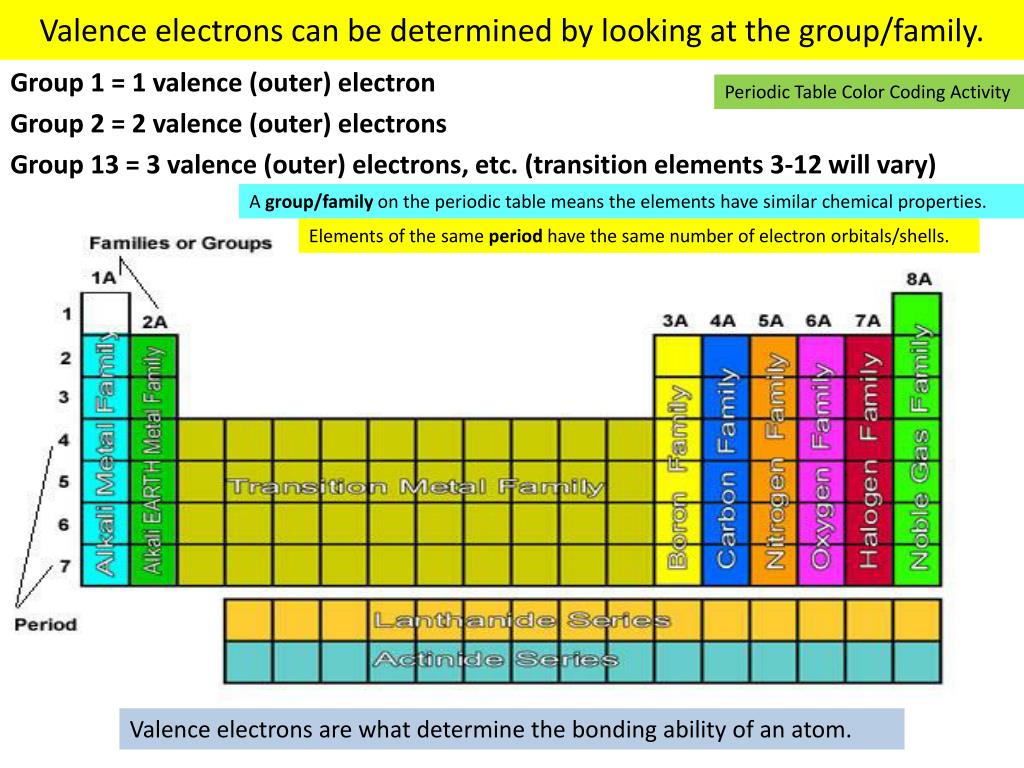
More than one electron), the energies of the subshells are no longer the same because However, for a multielectron atom or ion (an atom or ion containing a nucleus and Hydrogen-like electron system, the energies of the subshells depend only on theġs < 2s = 2p <3s = 3p = 3d < 4s = 4p = 4d = 4f <. R being directly proportional to n 2 where n is the principal quantum number. Given electron shell as E being inversely proportional to n 2 and the radius of the shell as The Bohr theory for a hydrogen-like atom or ion (an atom or ion containing a nucleusĪnd only one electron, e.g., H, He +, Li 2+ and Be 3+) gives the energy of the electron in a
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
